 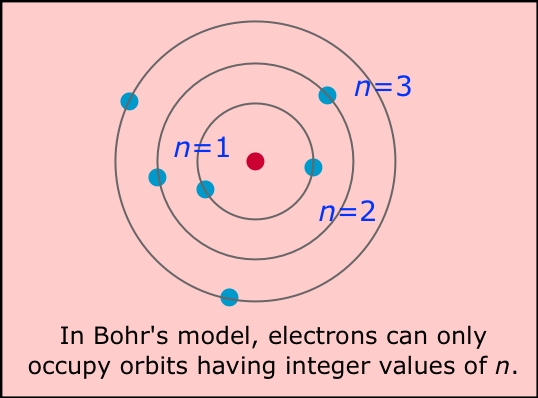
Thomson’s atomic model was later refined and replaced by Rutherford’s planetary model, which proposed that most of the mass and positive charge of the atom is concentrated in the nucleus, while the electrons orbit around it. The model also did not account for the existence of protons, which were later discovered by Ernest Rutherford and were found to be located in the atomic nucleus.The model did not account for the fact that electrons have a wave-like nature, which was later revealed by quantum mechanics.The model could not explain the existence of isotopes, which are atoms of the same element that have a different number of neutrons in their nuclei.It could not explain the phenomenon of radioactivity.It could not explain the observed phenomena of atomic spectra.It could not explain the stability of atoms, as the negatively charged electrons should have been attracted to the positively charged soup and collapsed into the center.Thomson’s model was able to explain some previously unexplained phenomena, such as the fact that atoms are electrically neutral, and that the ratio of the charge of an electron to its mass is the same for all elements.ġ.2.1 Limitations of J.J Thomson’s Model: He proposed that atoms were neutral because the negative charges of the electrons were balanced out by an equal amount of positive charge in the soup.  
He had discovered the electron, a subatomic particle while conducting experiments on cathode rays. In this model, Thomson proposed that atoms were composed of negatively charged electrons embedded in a positively charged “soup” or “pudding”. Thomson’s atomic model, also known as the “plum pudding” model, was proposed in 1904.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
